Data visuals 2017
Supplementary notes for CJ Brown’s talks on dataviz in 2017 for Griffith University’s honours students and the UQ Winterschool in Bioinformatics.
Skip to the quiz
Structure of this talk
- Tools for dataviz
- Eleven principles for effective dataviz
- Breaking the rules
- Change the world
The ideal dataviz tool
- Sensible and clean defaults
- Fast and convenient production of common graphics
- Convenient to plot statistical models
- Flexible enough to realize our creative thoughts
R integrates different tools
- Data merging
- Maps
- Graphs
- Analysis
- Word processing
- Presentations (including this one)
R is a flexible tool
- Flexibility is also it’s weakness
- Hard to start
- So many options to do the same thing
- Steep learning curve
- Often combine with other tools
Graphics packages in R
The dominant options are the base graphics R comes shipped with and the
ggplot2 package.
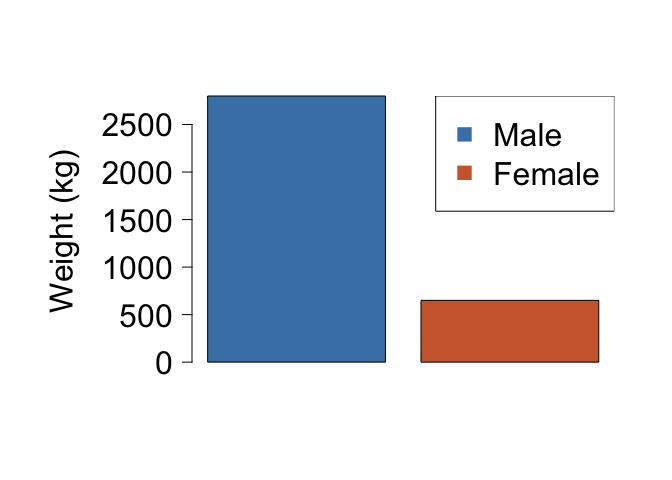
Plot your data
Make your own Datasaurus dozen
The datasaurus is a great example of why you should view your data, invented by Alberto Cairo. See Steph Locke’s code and package on github for making this in R.
library(datasauRus)
datnames <- rev(unique(datasaurus_dozen$dataset))
nlevels <- length(datnames)
for (i in 1:nlevels){
i <- which(datasaurus_dozen$dataset == datnames[i])
plot(datasaurus_dozen$x[i], datasaurus_dozen$y[i],
xlab = "x", ylab = "y", las = 1)
Sys.sleep(1)
}

Convince yourself that the mean, sd and correlation is the same in all of these plots:
library(dplyr)
datasaurus_dozen %>% group_by(dataset) %>%
summarize(meanx = mean(x), meany = mean(y),
sdx = sd(x), sdy = sd(y),
corr = cor(x,y))
## # A tibble: 13 × 6
## dataset meanx meany sdx sdy corr
## <chr> <dbl> <dbl> <dbl> <dbl> <dbl>
## 1 away 54.26610 47.83472 16.76982 26.93974 -0.06412835
## 2 bullseye 54.26873 47.83082 16.76924 26.93573 -0.06858639
## 3 circle 54.26732 47.83772 16.76001 26.93004 -0.06834336
## 4 dino 54.26327 47.83225 16.76514 26.93540 -0.06447185
## 5 dots 54.26030 47.83983 16.76774 26.93019 -0.06034144
## 6 h_lines 54.26144 47.83025 16.76590 26.93988 -0.06171484
## 7 high_lines 54.26881 47.83545 16.76670 26.94000 -0.06850422
## 8 slant_down 54.26785 47.83590 16.76676 26.93610 -0.06897974
## 9 slant_up 54.26588 47.83150 16.76885 26.93861 -0.06860921
## 10 star 54.26734 47.83955 16.76896 26.93027 -0.06296110
## 11 v_lines 54.26993 47.83699 16.76996 26.93768 -0.06944557
## 12 wide_lines 54.26692 47.83160 16.77000 26.93790 -0.06657523
## 13 x_shape 54.26015 47.83972 16.76996 26.93000 -0.06558334
We can also save these as .png images to make a .gif image (see also here)
for (ilvs in 1:nlevels){
i <- which(datasaurus_dozen$dataset == datnames[ilvs])
thiscol <- ifelse(datnames[ilvs] == "dino", "darkseagreen", "grey20")
png(filename = paste0("datasaurus/",datnames[ilvs],".png"))
plot(datasaurus_dozen$x[i], datasaurus_dozen$y[i],
xlab = "x", ylab = "y", las = 1,
xlim = c(10, 105), ylim = c(-5, 105), col = thiscol, pch = 16)
dev.off()
}
Clarity not simplicity
I give the example of the famout ‘hockey stick’ graph of Northern Hemisphere temperatures.
Dataviz are models
Any visualization is a model
Alberto Cairo 2016
Different viz models for the same data
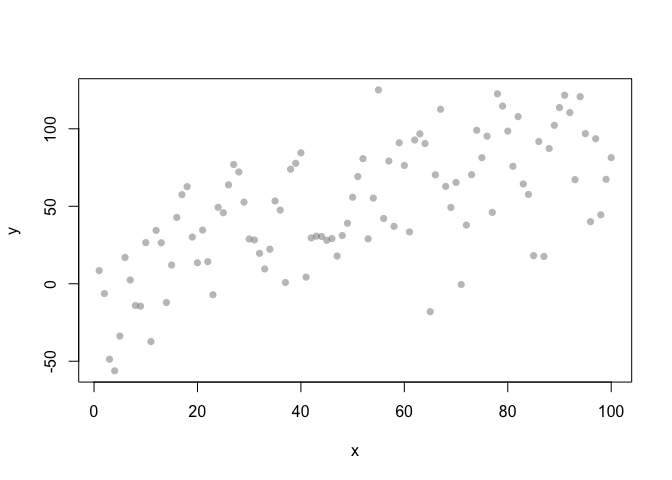
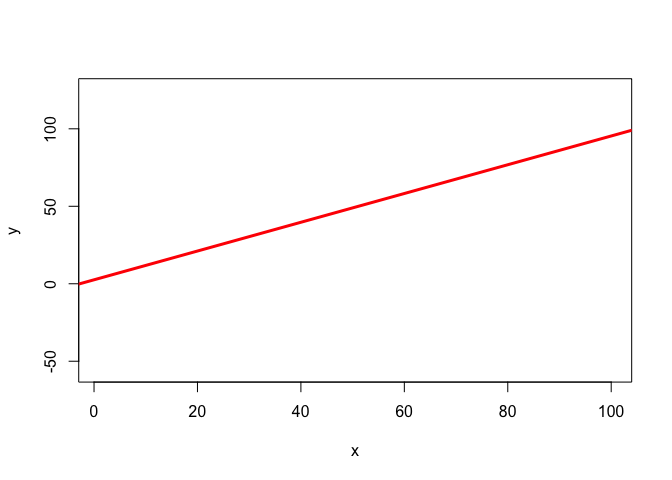
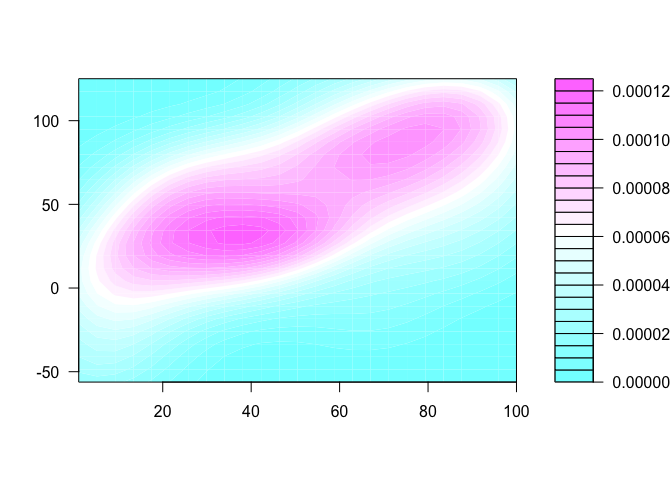
Three ways of visualising the same x-y data
Each of these graphs of the same data has a slightly different interpretation.
x <- 1:100
y <- x + rnorm(100, sd=30)
plot(x,y, pch = 16, col = grey(0.5, 0.5))
mod1 <- lm(y ~ x)
plot(x,y, col = 'white')
abline(mod1, lwd = 3, col = 'red')
library(MASS)
filled.contour(kde2d(x,y), scale = F)
plot(x,y, pch = 16, col = grey(0.5, 0.5))
abline(mod1, lwd = 3, col = 'red')
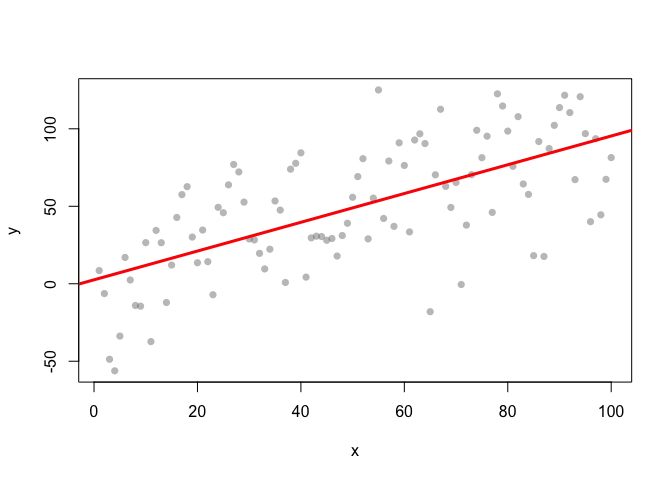
Models help clarify complex data-sets
Effect size often has to been seen to be understood
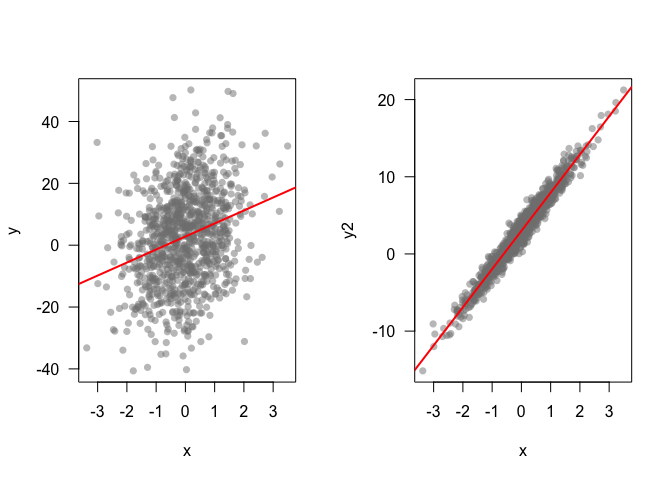
When doing confirmatory analysis, we might want to know how strong an effect is. Data viz is very useful for this task. Lets compare two datasets that have similar p-values, but very different effect sizes
set.seed(42)
x <- rnorm(1000)
set.seed(420)
y <- 5*x + 3 + rnorm(1000, sd = 15)
set.seed(420)
y2 <- 5*x + 3 + rnorm(1000, sd = 1)
mod1 <- lm(y ~ x)
mod2 <- lm(y2 ~ x)
#Compare the pvalues on the slopes
summary(mod1)
##
## Call:
## lm(formula = y ~ x)
##
## Residuals:
## Min 1Q Median 3Q Max
## -43.201 -10.330 0.395 9.634 46.694
##
## Coefficients:
## Estimate Std. Error t value Pr(>|t|)
## (Intercept) 2.8054 0.4614 6.080 1.71e-09 ***
## x 4.2096 0.4603 9.145 < 2e-16 ***
## ---
## Signif. codes: 0 '***' 0.001 '**' 0.01 '*' 0.05 '.' 0.1 ' ' 1
##
## Residual standard error: 14.59 on 998 degrees of freedom
## Multiple R-squared: 0.07732, Adjusted R-squared: 0.07639
## F-statistic: 83.63 on 1 and 998 DF, p-value: < 2.2e-16
summary(mod2)
##
## Call:
## lm(formula = y2 ~ x)
##
## Residuals:
## Min 1Q Median 3Q Max
## -2.88004 -0.68868 0.02634 0.64229 3.11291
##
## Coefficients:
## Estimate Std. Error t value Pr(>|t|)
## (Intercept) 2.98703 0.03076 97.11 <2e-16 ***
## x 4.94731 0.03069 161.21 <2e-16 ***
## ---
## Signif. codes: 0 '***' 0.001 '**' 0.01 '*' 0.05 '.' 0.1 ' ' 1
##
## Residual standard error: 0.9724 on 998 degrees of freedom
## Multiple R-squared: 0.963, Adjusted R-squared: 0.963
## F-statistic: 2.599e+04 on 1 and 998 DF, p-value: < 2.2e-16
par(mfrow = c(1,2))
plot(x,y, pch = 16, col = grey(0.5,0.5), las = 1)
abline(mod1, lwd = 2, col = 'red')
plot(x,y2, pch = 16, col = grey(0.5,0.5), las = 1)
abline(mod2, lwd = 2, col = 'red')

Superplots
Andrew Gelman coined the term superplots for plotting different models on multiple panels of a graph so you can visually compare them.
For instance, say we have several time-series and we want to know if they deviate from each other signficantly. An easy way to compare them is to fit splines to each time-series and then just plot them next to each other, with SEs. Then we can compare visually for ‘signficant’ differences.
Here’s some code to simulate three made-up series. The first two have the same trend, but different observation errors, the third has a different trend:
tmax <- 50
drift <- c(10, -5)
sd <- 40
sdobs <- 200
set.seed(5)
yzero <- cumsum(rnorm(n=tmax, mean=drift[1], sd=sd))
y1 <- yzero + rnorm(n = tmax, mean = 0, sd = sdobs)
y2 <- yzero + rnorm(n = tmax, mean = 0, sd = sdobs)
y3 <- cumsum(rnorm(n=tmax, mean=drift[2], sd=sd)) +
rnorm(n = tmax, mean = 0, sd = sdobs)
dat <- data.frame(ts = rep(letters[1:3], each = tmax), x = rep(1:tmax, 3), y = c(y1, y2, y3))
We can easily plot these three series using ggplot2 and automatically
add a spline.
library(ggplot2)
ggplot(dat, aes(x = x, y = y)) +
geom_point() +
facet_grid(.~ts) +
stat_smooth(method = "loess", se = TRUE) +
theme(axis.text = element_text(size=14),
axis.title = element_text(size=16,face="bold"),
strip.text.x = element_text(size = 16),
panel.background = element_rect(fill = 'white', colour = 'white'))

Length is most accurate
Ways of comparing data in order from most accurate (top) to more generic (bottom).

Comparing volume and area
Compare these. Note that if we compare circles we should use area, not the radius or diameter to scale their size.
n <- c(10, 5)
barplot(n, col = 'skyblue', xaxt = 'n', yaxt = 'n')
rad1 <- 1
area1 <- pi*(rad1^2)
area2 <- area1/2
rad2 <- sqrt(area2/pi)
par(mfrow = c(1,2), mar = c(0,0,0,0))
pie(1, col = 'skyblue', labels = NA, border = NA, radius = rad1)
pie(1, col = 'skyblue', labels = NA, border = NA, radius = rad2)

Exploration of data
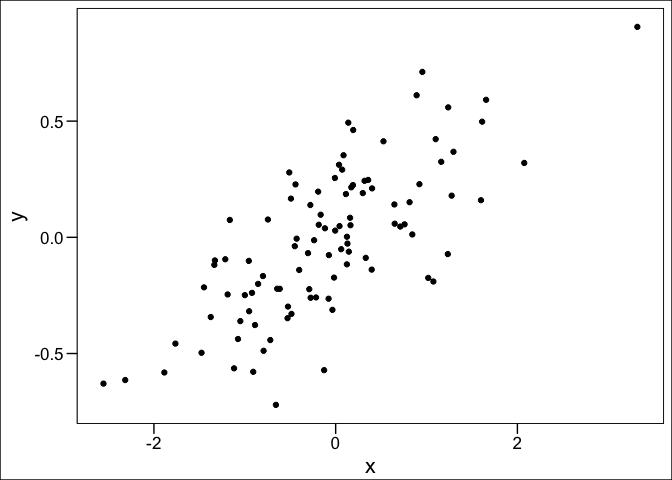
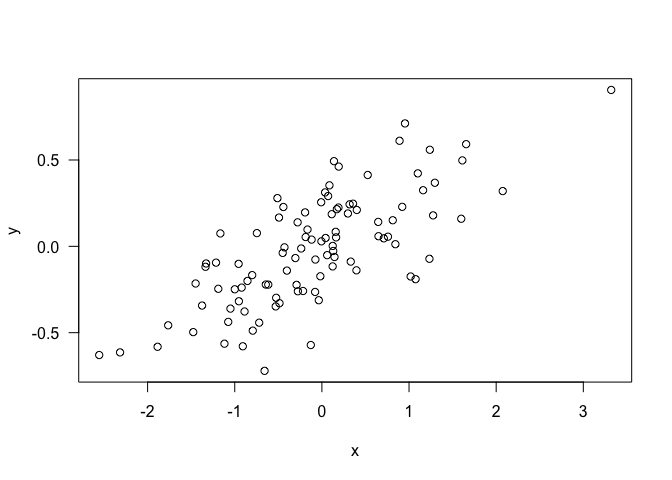
Let’s create a point cloud to demonstrate some data exploration techniques
set.seed(42)
x <- rnorm(1000)
y <- 5*x + 3 + rnorm(1000, sd = 15)
plot(x,y, pch = 16, col = grey(0.5,0.5), las = 1)
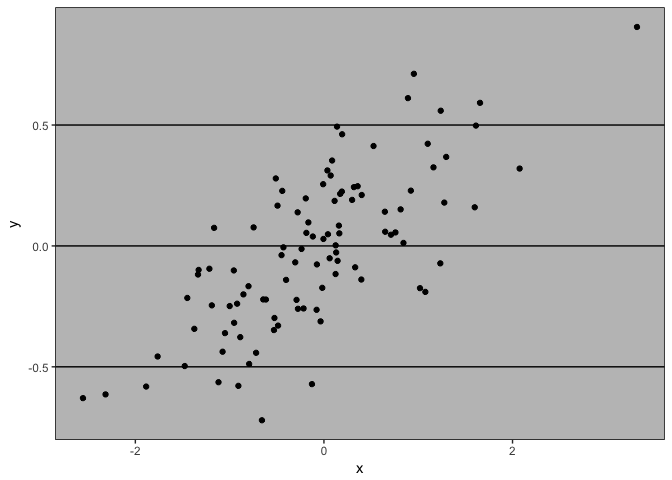
Can’t see alot here. A linear model might help us explore if there is any trend going on:
mod1 <- lm(y ~ x)
plot(x,y, pch = 16, col = grey(0.5,0.5), las = 1)
abline(mod1, lwd = 2, col = 'red')
xnew <- seq(min(x), max(x), length.out = 100)
pmod <- predict(mod1, newdata =data.frame(x=xnew), se = T)
lines(xnew, pmod$fit + pmod$se.fit, lwd = 2, col = 'red', lty = 2)
lines(xnew, pmod$fit - pmod$se.fit, lwd = 2, col = 'red', lty = 2)
What about identifying extreme points, that may be worth investigating further? We can pick out points that are greater than 2SDs from the trend:
modresid <- resid(mod1)
sd2 <- sd(modresid)*2
ipt <- which(abs(modresid) > sd2)
plot(x,y, pch = 16, col = grey(0.5,0.5), las = 1)
abline(mod1, lwd = 2, col = 'red')
points(x[ipt], y[ipt], pch = 16, col = rgb(1,0,0, 0.6))
Effect size
Don’t waste digital ink
Plots with less ‘add-ons’ tend to communicate the key message more clearly. For instance, just like excel plots dont:
x <- rnorm(100)
dat <- data.frame(x = x, y = 0.25*x + rnorm(100, sd = 0.2))
library(ggplot2)
library(ggthemes)
ggplot(dat, aes(x = x, y = y)) + geom_point() +
theme_excel() + theme(axis.text=element_text(size=20),
axis.title=element_text(size=20))
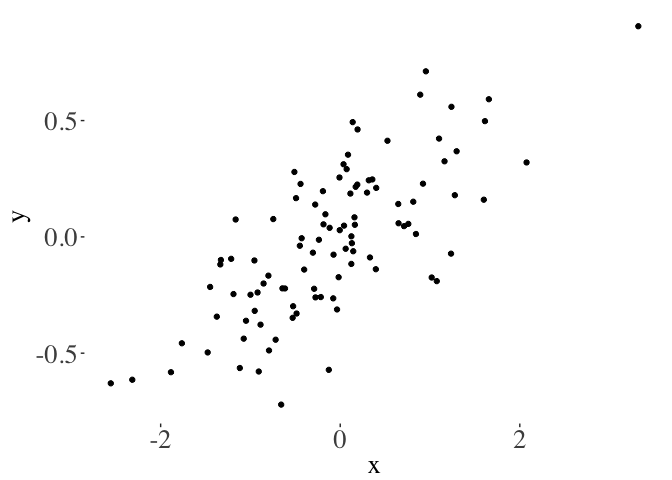
You can get additional themes for ggplot2 using this excellent package. A cleaner view:
ggplot(dat, aes(x = x, y = y)) + geom_point() +
theme_base() + theme(axis.text=element_text(size=20),
axis.title=element_text(size=20))
Or simply:
plot(dat$x, dat$y, xlab = "x", ylab = "y", las = 1)
A good principle is to not use ‘ink’ on figures that isn’t needed to communicate your message. Tufte takes the ‘less ink’ philosophy to the extreme:
ggplot(dat, aes(x = x, y = y)) + geom_point() +
theme_tufte() + theme(axis.text=element_text(size=20),
axis.title=element_text(size=20))
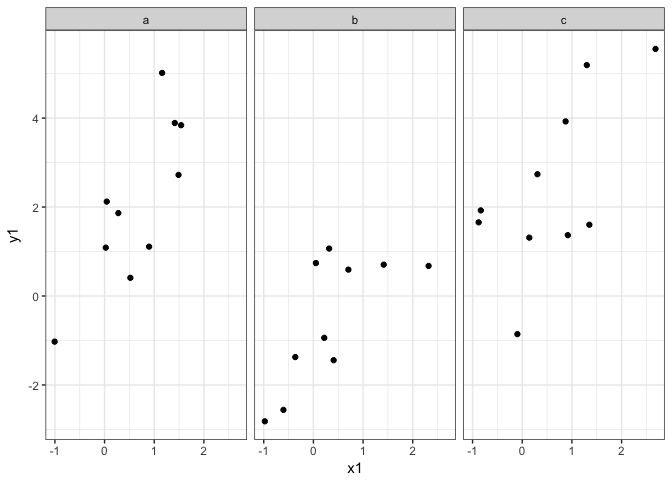
When is ggplot2 appropriate, or when should I use base R?
In general I think ggplot2 is appropriate for problems of intermediate complexity. Like this:
 Base R is great if you just want to plot a barplot quickly, or do an x-y
plot. ggplot2 comes into its own for slight more complex plots, like
having multiple panels for different groups or colouring lines by a 3rd
factor. But once you move to really complex plots, like overlaying a
subplot on a map, ggplot2 becomes very difficult, if not impossible. At
that point it is better to move back to Base R. ggplot2 can also get
very fiddly if you are very specific about your plots, like having
certain colours, or the labels in a certain way.
Base R is great if you just want to plot a barplot quickly, or do an x-y
plot. ggplot2 comes into its own for slight more complex plots, like
having multiple panels for different groups or colouring lines by a 3rd
factor. But once you move to really complex plots, like overlaying a
subplot on a map, ggplot2 becomes very difficult, if not impossible. At
that point it is better to move back to Base R. ggplot2 can also get
very fiddly if you are very specific about your plots, like having
certain colours, or the labels in a certain way.
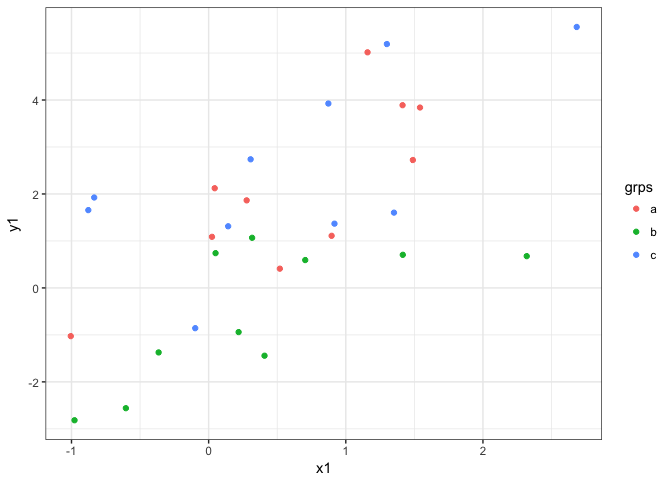
As an example, ggplot2 is great for data like this:
x1 <- rnorm(30)
grps <- letters[c(rep(1, 10), rep(2, 10), rep(3, 10))]
y1 <- x1 + c(rep(1, 10), rep(-1, 10), rep(2, 10)) + rnorm(30)
dat <- data.frame(x = x1, grps = grps, y = y1)
head(dat)
## x grps y
## 1 0.19033984 a 6.159889e-01
## 2 -0.07173877 a -7.577584e-05
## 3 -0.00285171 a -1.003277e+00
## 4 -1.10821896 a -7.302877e-01
## 5 0.93519177 a 2.662337e+00
## 6 -1.48583945 a -1.588023e+00
ggplot(dat, aes(x = x1, y = y1, color = grps)) +
geom_point() + theme_bw()
It is also pretty handy for faceting:
ggplot(dat, aes(x = x1, y = y1)) +
geom_point() + facet_wrap(~grps)+
theme_bw()
The key with ggplot2 is to have your data in a data-frame.
In reality both ggplot2 and base R graphics are worth learning, but I would start with learning the basics of base R graphics and then move onto ggplot2 if you want to quickly plot lots of structured data-sets.
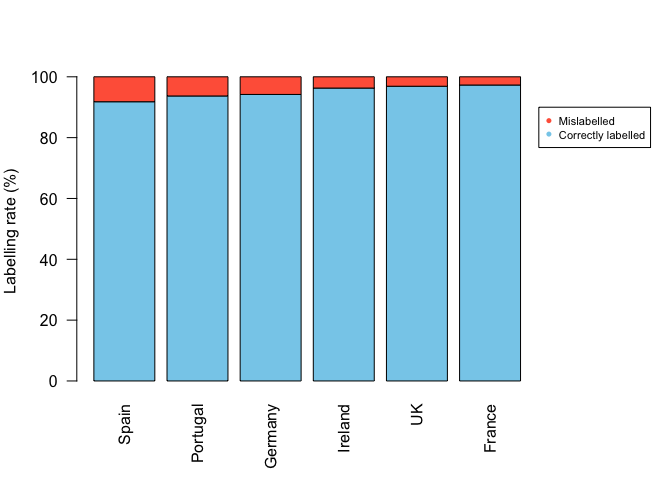
Pie graphs vs bar graphs
In Mariani et al. they plot rates of seafood fraud by several European countries. While its a foundational study that establishes improvement in the accuracy of food labelling, their graphics could be improved in several ways.
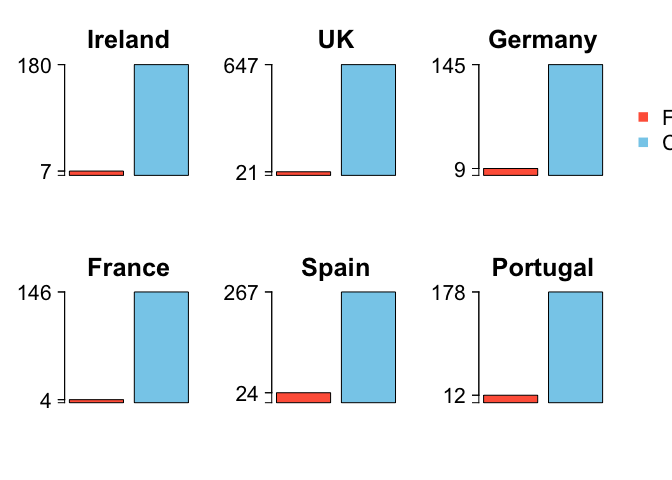
First they use perspective pies. This makes it incredibly hard to compare the two groups (fish that are labelled/mislabelled). Humans are very bad at comparing angles and pretty bad at comparing areas. With the perspective you can’t even compare the areas properly. They do provide the raw numbers, but then, why bother with the pies? Note that the % pies misrepresent the data slightly because the % figures are actually odds ratios (mis-labels / correct labels), rather than percent (mis-labeels / total samples). Second the pies are coloured red/green, which will be hard for red-green colourblind people to see. Third, they have coloured land blue on their map, so it appears to be ocean at first look. Fourth, the map is not really neccessary. There are no spatial patterns going on that the authors want to draw attention to. I guess having a map does emphasize that the study is in Europe. Finally, the size of each pie is scaled to the sample size, but the scale bar for the sample size shows a sample of only 30, whereas most of their data are for much larger samples sizes (>200). Do you get the impression from the pies that the UK has 668 samples, whereas Ireland only has 187? Therefore, from this graphic we have no idea what sample size was used in each country.
In fact, all the numbers that are difficult to interpret in the figure are very nicely presented in Table 1.
Below is a start at improving the presentation. For instance, you could do a simple bar chart, ordering by rate of mislabelling.
cnames <- c('Ireland' ,'UK','Germany','France','Spain','Portugal')
corrlab <- c(180, 647, 145, 146, 267, 178)
mislab <- c(7, 21, 9, 4, 24, 12)
misrate <- 100*signif(mislab / (corrlab + mislab),2)
corrrate <- 100 - misrate
ord <- order(misrate, decreasing = T)
y <- rbind(corrrate, misrate)
par(mar = c(5,4,4,7))
barplot(y[,ord], names.arg = cnames[ord], col = c('skyblue','tomato'), ylab = 'Labelling rate (%)', las = 2)
legend(x=7.5, y = 90, legend = c("Mislabelled", "Correctly labelled"), pch = 16, col = c('tomato','skyblue'), xpd = NA, cex = 0.7)
You could add another subfigure to this, showing the rate by different species too.
The barplot doesn’t communicate the sample size, but then that is probably not the main point. The sample sizes are probably best reported in the table
If we felt the map was essential, then putting barcharts on it would be more informative. It is not that easy to add barcharts ontop of an existing map in R, so I would recommend creating the barcharts seperately, then adding them on in Illustrator or Powerpoint.
We can make a basic map like this:
library(maps)
library(maptools)
map('world', xlim = c(-20, 20), ylim = c(35, 60), col = 'grey', fill = T)

Then create some nice barcharts. We write a loop so we get one barchart for each country.
nc <- length(cnames)
par(mfrow = c(2,3), oma = c(1,1,1,3))
for (i in 1:nc){
y <- c(mislab[i], corrlab[i])
barplot(y, names.arg = '', las = 2, col = c('tomato','skyblue'), ylim = c(0, corrlab[i]), main = cnames[i], cex.main = 2.4, yaxt = 'n')
byy <- signif(max(y),2)/3
yat <- c(0, min(y), max(y))
axis(2, at = yat, las = 2, cex.axis = 2, labels = F)
axis(2, at = yat[2:3], las = 2, cex.axis = 2, labels = T)
}
legend(x = 2.8, y = 500, legend = c('Fraud', 'Correct'), pch = 15, col = c('tomato','skyblue'), xpd = NA, cex = 2, bty = 'n')
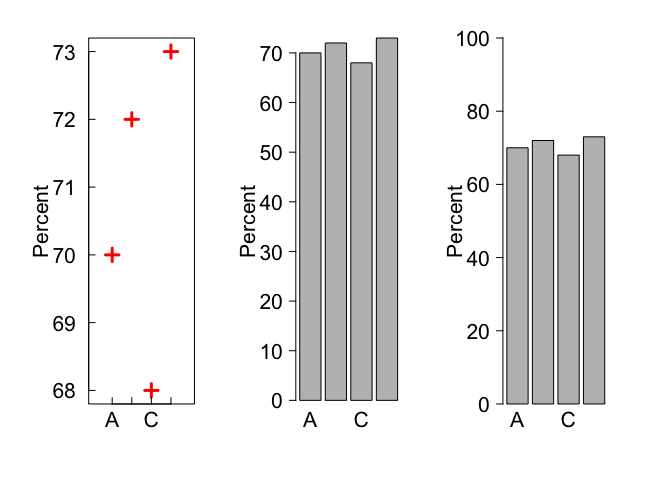
Scaling matters
It can be misleading to present % and proportion data on axes that are not scaled 0 - 100%. For instance, compare these three graphs:
y <- c(70, 72, 68, 73)
x <- 1:4
xnams <- LETTERS[1:4]
par(mfrow = c(1,3), oma = c(1,1,1,3), mar = c(5,6,2,2))
plot(x,y, pch = 3, cex = 2, las = 1, xaxt = 'n', xlab = '', ylab = 'Percent', cex.axis = 2, cex.lab = 2, tcl = 0.5, xlim = c(0, 5), col = 'red', lwd = 3)
axis(1, at = x, labels = xnams, cex.axis = 2, tcl = 0.5)
barplot(y, names.arg = xnams, las = 1, cex.axis = 2, cex.lab = 2, cex.names = 2, ylab = 'Percent')
barplot(y, names.arg = xnams, las = 1, cex.axis = 2, cex.lab = 2, cex.names = 2, ylab = 'Percent', ylim = c(0, 100))
Interpreting rates
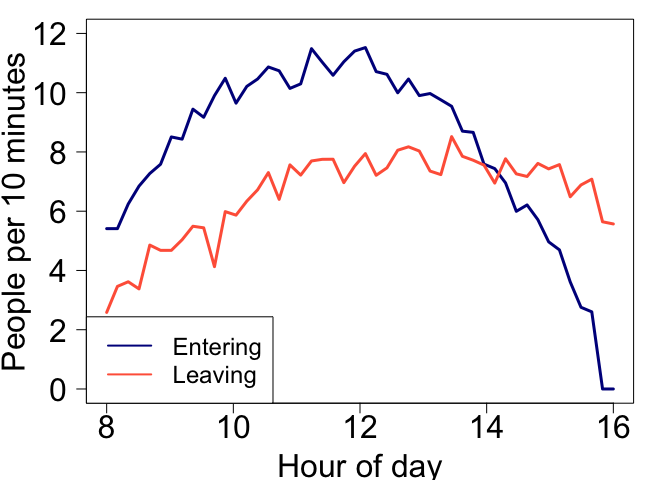
The units you use affect how people interpret your graph. People are bad at interpreting rates, we just can’t get our heads around accumulation very well. Here is a numerical example. Check out the below figure and ask yourself:
At what time is the number of people in the shopping centre declining?
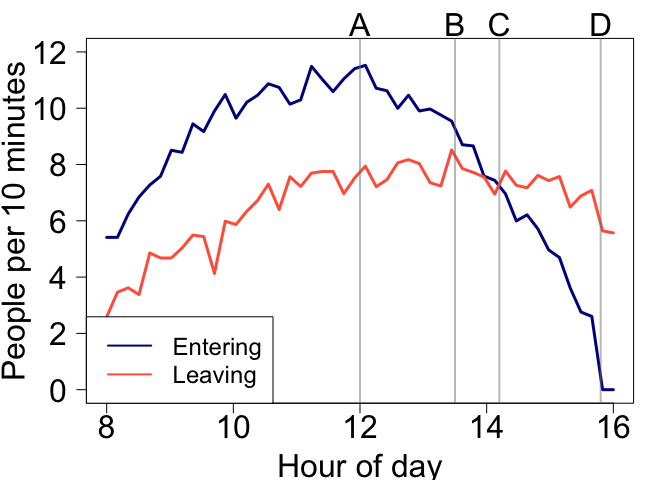
Would you say it is at point A, B, C or D?
Before you proceed with code below, take the poll:
Here is how we made the figure and generated the data:
par(mar = c(4,4.5,2,2), mgp = c(3,1,0))
plot(times, inrate_err, type = 'l', xlab = 'Hour of day', ylab = 'People per 10 minutes', las = 1, cex.axis = 2, lwd = 3, col = 'darkblue', cex.lab = 2, ylim = c(0, 12))
lines(times, outrate_err, lwd = 3, col = 'tomato')
abline(v = 12, lwd = 2, col = grey(0.5,0.5))
text(12, 13, 'A', xpd = NA, cex = 2)
abline(v = 13.5, lwd = 2, col = grey(0.5,0.5))
text(13.5, 13, 'B', xpd = NA, cex = 2)
abline(v = 14.2, lwd = 2, col = grey(0.5,0.5))
text(14.2, 13, 'C', xpd = NA, cex = 2)
abline(v = 15.8, lwd = 2, col = grey(0.5,0.5))
text(15.8, 13, 'D', xpd = NA, cex = 2)
legend('bottomleft', legend = c('Entering', 'Leaving'), lwd = 2, col = c('darkblue','tomato'), cex = 1.5, xpd = NA)
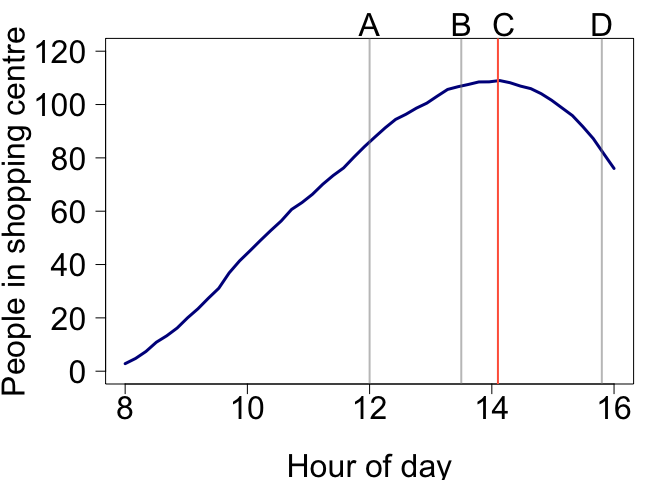
Let’s plot the total number of people:
par(mar = c(5,5.5,2,2), mgp = c(4,1,0))
plot(times, cumsum(inrate_err) - cumsum(outrate_err), type = 'l', xlab = 'Hour of day', ylab = 'People in shopping centre', las = 1, cex.axis = 2, lwd = 3, col = 'darkblue', cex.lab = 2, ylim = c(0, 120))
abline(v = 12, lwd = 2, col = grey(0.5,0.5))
text(12, 130, 'A', xpd = NA, cex = 2)
abline(v = 13.5, lwd = 2, col = grey(0.5,0.5))
text(13.5, 130, 'B', xpd = NA, cex = 2)
abline(v = 14.1, lwd = 2, col = 'tomato')
text(14.2, 130, 'C', xpd = NA, cex = 2)
abline(v = 15.8, lwd = 2, col = grey(0.5,0.5))
text(15.8, 130, 'D', xpd = NA, cex = 2)
Hopefully the answer is obvious now. So the right scales can help make interpretation much easier.
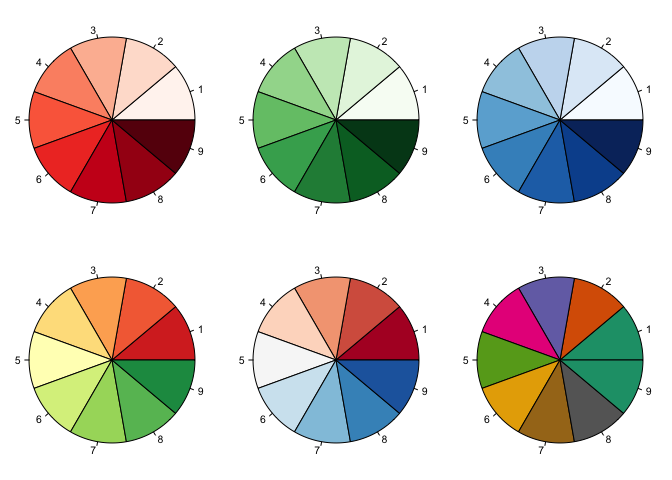
Choosing colour scales
Alot of thought should go into choosing colour scales for graphs for
instance- will it print ok? will colour blind people be able to see
this? does the scale create artificial visual breaks in the data?
Luckily there is a package to help you make the right decision for a
colour scale, it is called RColorBrewer. Check out colorbrewer.org for
a helpful interactive web interface for choosing colours.
First let’s load some sea surface temperature data as a raster:
library(raster)
r <- raster("MeanAVHRRSST")
library(RColorBrewer)
par(mfrow = c(1,2))
plot(r, col = rev(brewer.pal(11, "Spectral")), asp = NA)
plot(r, col = brewer.pal(11, "Purples"), asp = NA)
## Warning in brewer.pal(11, "Purples"): n too large, allowed maximum for palette Purples is 9
## Returning the palette you asked for with that many colors
Using red-green palettes makes it hard for colour blind people. Also, using a diverging palette makes it look like there is something important about the middle point (yellow). A better palette to use would be one of the sequential ones, “Purples” shown here.
To make it easier to understand, let’s look at these again as contour plots. I will use a more appropriate diverging palette to the red-green one though.
z <- matrix(rep(1:10, 10), nrow = 10)
filled.contour(z, col = brewer.pal(9, 'Reds'), nlevels = 10)
filled.contour(z, col = brewer.pal(9, 'RdBu'), nlevels = 10)

Notice the diverging pallette creates an artificial split at yellow
One of the only legitimate uses for pie graphs (I think) is visualising the colour scales. Here is how:
reds <- brewer.pal(9, 'Reds')
greens <- brewer.pal(9, 'Greens')
blues <- brewer.pal(9, 'Blues')
rdylgn <- brewer.pal(9, 'RdYlGn')
rdbu <- brewer.pal(9, 'RdBu')
dark2 <- brewer.pal(8, 'Dark2')
par(mfrow = c(2,3), mar = c(0,0,0,0), oma = c(0,0,0,0))
pie(rep(1, 9), col = reds)
pie(rep(1, 9), col = greens)
pie(rep(1, 9), col = blues)
pie(rep(1, 9), col = rdylgn)
pie(rep(1, 9), col = rdbu)
pie(rep(1, 9), col = dark2)
Breaking the rules - stick with convention
Sometimes you might want to bend or break the principles above in order to stick with convention. This is useful if people are used to viewing data in a particular way. For instance, geneticists are fond of pie graphs in haplotype networks. Here is some code I modified from here to make one (note that in this data-set there is no haplotype diversity so we artificially mix them up so you can see the pies here):
library(pegas)
library(RColorBrewer)
data(woodmouse)
x <- woodmouse[sample(15, size = 110, replace = TRUE), ]
h <- haplotype(x)
net <- haploNet(h)
mixed_up <- rep(letters[1:5], each=22)
ind.hap2 <- with(
utils::stack(setNames(attr(h, "index"), rownames(h))),
table(hap=ind, pop = mixed_up[values])
)
plot(net, size = attr(net, "freq"), scale.ratio = 2, cex = 0.8, pie = ind.hap2, bg = brewer.pal(8, "Dark2"))
legend("bottomright", colnames(ind.hap2), col = brewer.pal(8, "Dark2"), pch=20)

Interactive dataviz
Interactive visuals can overcome the dilemma of having too much complexity to show, but also wanting the viewer to explore the details. For instance, you could use the dygraphs package to zoom in on a date range.
We will get some recent data on wave heights from QLD government. First we load the data directly from the web and process it to correctly label dates and times, then just select the Signficant wave height variable at Tweed Heads.
waves <-read.csv(url("http://www.ehp.qld.gov.au/data-sets/waves/wave-7dayopdata.csv?timestamp=2017-07-05EST21-06-50"), skip = 1)
waves$time <- as.POSIXct(strptime(as.character(waves$DateTime), format = "%Y-%m-%dT%H:%M:%S"))
waves2 <- subset(waves, (Hsig > 0) & (Site == "Tweed Heads"))[,c("time", "Hsig")]
waves_xts <- xts::xts(waves2$Hsig, order.by = waves2$time)
Now have our data, we can look at it using the dygraphs package. We will do two things: add a date range selector and also add a ‘roll’ which will smooth over a certain period of our choosing:
library(dygraphs)
dygraph(waves_xts, main = "Significant wave height - Tweed Heads") %>%
dyRoller(rollPeriod = 50) %>%
dyRangeSelector()
I haven’t put it up on the webpage here, but have a go yourself.
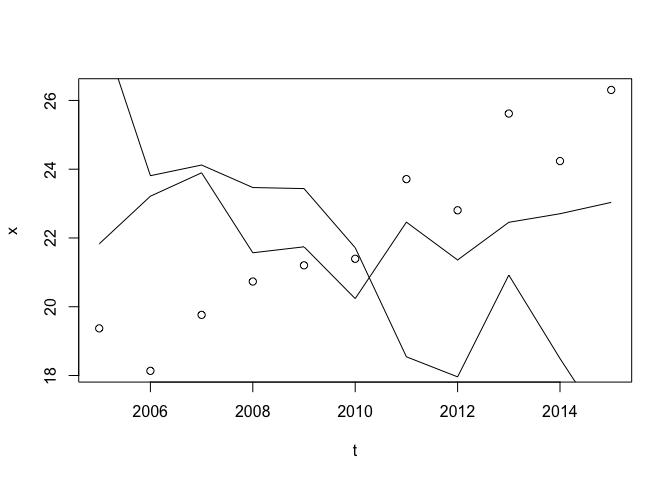
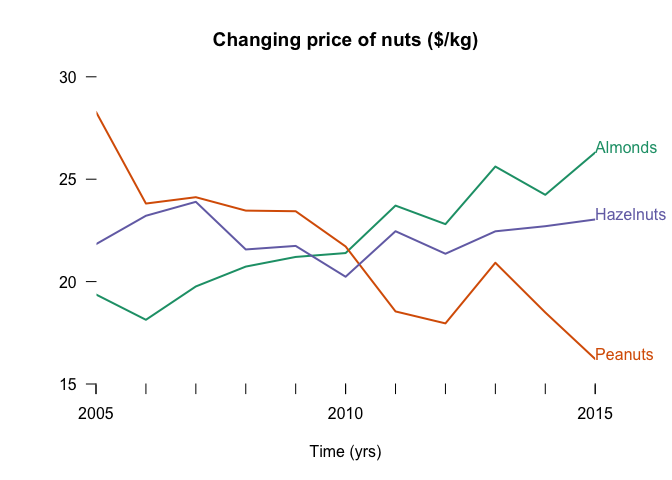
Anatomy of a simple chart
The construction of a simple chart in R can be a surprisingly long piece of code. Here is an example to get you started. Don’t be afraid to experiment!
# --------------- #
# Make some data
# --------------- #
set.seed(42)
n <- 11
x <- rnorm(n, mean = seq(18, 25, length.out = n))
y <- rnorm(n, mean =seq(26, 18, length.out = n))
z <- rnorm(n, mean = 22)
t <- 2005:(2005+n-1)
datnames <- c('Almonds', 'Peanuts', 'Hazelnuts')
plot(t, x)
lines(t, y)
lines(t, z)

Which look terrible. Let’s build a better chart.
# Package for colours
library(RColorBrewer)
#Set axis limits
ymax <- 30
ylim <- c(15, ymax)
xlim <- c(min(t), max(t))
#Define colours
cols <- brewer.pal(3, 'Dark2')
#Parameters for plotting
lwd <- 2
xlabels <- seq(min(t), max(t), by = 5)
ylabels <- seq(0, ymax, by = 5)
#Set the window params
par(mar = c(5,5,4,4))
#Build the plot
plot(t, x, type = 'l', bty = 'n', xaxt = 'n', yaxt = 'n',
ylim = ylim, xlim = xlim, lwd = lwd, col = cols[1],
xaxs = 'i', yaxs = 'i',
xlab = 'Time (yrs)',
ylab = '',
main = 'Changing price of nuts ($/kg)')
#Add more lines
lines(t, y, lwd = lwd, col = cols[2])
lines(t, z, lwd = lwd, col = cols[3])
#Add labels to lines
text(t[n], x[n], datnames[1], adj = c(0, 0), xpd = NA, col = cols[1])
text(t[n], y[n], datnames[2], xpd = NA, adj = c(0, 0), col = cols[2])
text(t[n], z[n], datnames[3], xpd = NA, adj = c(0, 0), col = cols[3])
# Add custom axes
axis(1, col = 'white', col.ticks = 'black', labels = xlabels, at = xlabels)
axis(1, col = 'white', col.ticks = 'black', labels = NA, at = t)
axis(2, col = 'white', col.ticks = 'black', las =1, labels = ylabels, at = ylabels)

Resources and further reading
-
An infographic of chart types: Visual vocabulary